 However, the model assumed electrons in the shells didn't interact with each other and couldn't explain why electrons seemed to stack in an irregular manner. It also explained why the noble gases were inert and why atoms on the left side of the periodic table attract electrons, while those on the right side lose them. Bohr Model Schalenmodell Periodic Table Krypton Electron Shell, PNG, 1280x885px, Bohr Model, Area, Atom, Carbon, Chemical Element Download Free User kamikalbaba uploaded this CAFFè - Bohr Model Schalenmodell Periodic Table Krypton Electron Shell PNG PNG image on March 5, 2018, 11:00 am. 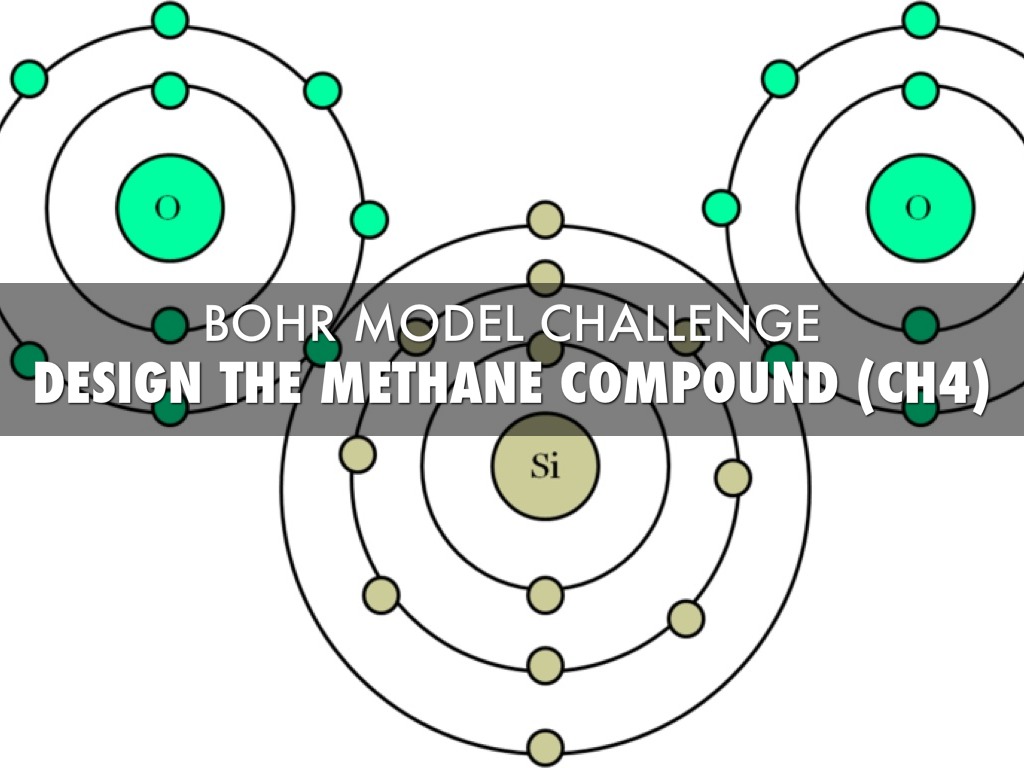
For example, the shell model explained why atoms got smaller moving across a period (row) of the periodic table, even though they had more protons and electrons. The model explained some of the atomic properties of heavier atoms, which had never been reproduced before. Thousands of new, high-quality pictures added every day. Thus, the Bohr model for heavier atoms described electron shells. Carbon Atom Bohr Model Vector Editable Stock Vector (Royalty Free) 1992619808 Shutterstock Find Carbon Atom Bohr Model Vector Editable stock images in HD and millions of other royalty-free stock photos, illustrations and vectors in the Shutterstock collection. Once the level was full, additional electrons would be bumped up to the next level. EDITORIAL VIDEO 20 Bohr Atomic Model Stock Photos & High Res Pictures Browse 20 bohr atomic model photos and images available, or start a new search to explore more photos and images. 1 That is, the Bohr effect refers to the shift in the oxygen dissociation curve. 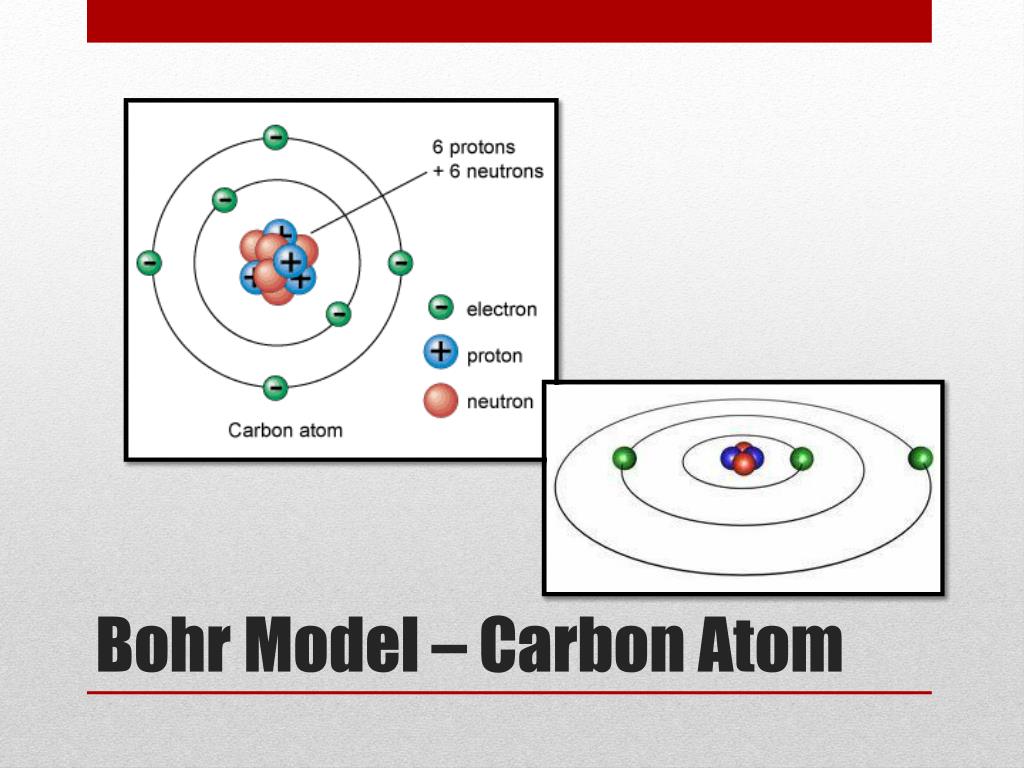
Hemoglobin 's oxygen binding affinity (see oxygenhaemoglobin dissociation curve) is inversely related both to acidity and to the concentration of carbon dioxide. This nucleus is surrounded by two-electron shells named K-shell and L-shell. Bohr believed each electron orbit could only hold a set number of electrons. The Bohr effect is a phenomenon first described in 1904 by the Danish physiologist Christian Bohr. Published By Vishal Goyal Last updated: DecemBohr model of Carbon atom - How to draw Carbon (C) Bohr-Rutherford diagram Home > Chemistry > Carbon Bohr model The Bohr Model of Carbon (C) has a nucleus that contains 6 neutrons and 6 protons. More electrons were required to cancel out the positive charge of all of these protons. In Bohr's model, the innermost shell can have a maximum of two electrons. 
Bohr found that an electron located away from the nucleus has more energy, and the electron which. It shows the atoms electrons in circular orbits at specific distances from the nucleus. 3 signifying In calcite, that one the carbon molecule and contains three oxygen. Bohr’s model consists of a small nucleus (positively charged) surrounded by negative electrons moving around the nucleus in orbits. Rutherford’s earlier model of the atom had also assumed that electrons moved in circular orbits around the nucleus and that the atom was held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electron.Heavier atoms contain more protons in the nucleus than the hydrogen atom. Rutherford explained the nucleus of an atom and Bohr modified that model into electrons and their energy levels. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. The electrons in the outermost shell (main energy level) of an atom these are the electrons involved in forming bonds. In 1913, a Danish physicist, Niels Bohr (1885–1962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. Where \(n_1\) and \(n_2\) are positive integers, \(n_2 > n_1\), and \( \Re \) the Rydberg constant, has a value of 1.09737 × 10 7 m −1. The solar system or planetary model of the atom was attractive to scientists because it was similar to something with which they were already familiar, namely the solar system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
